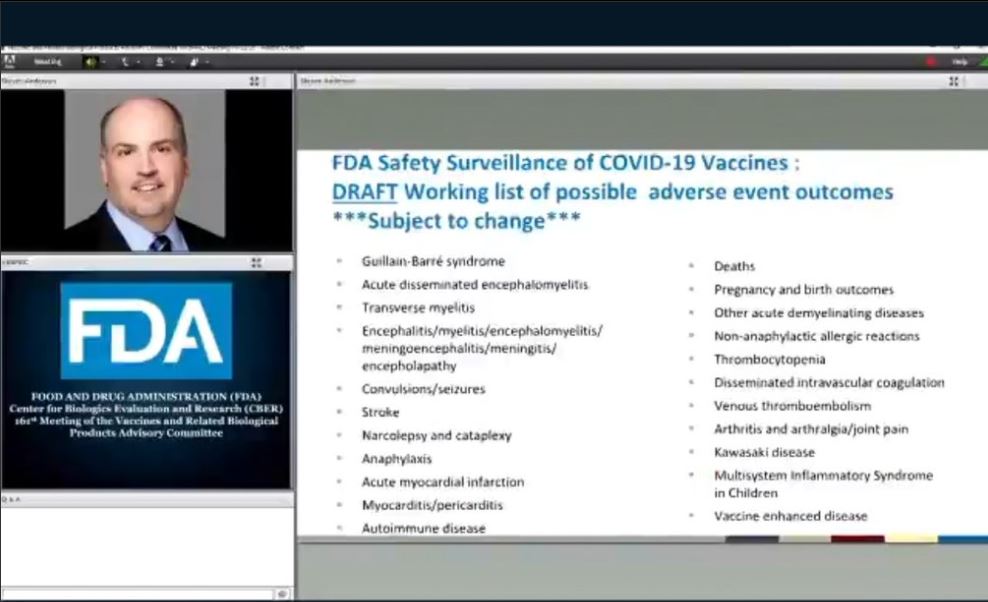 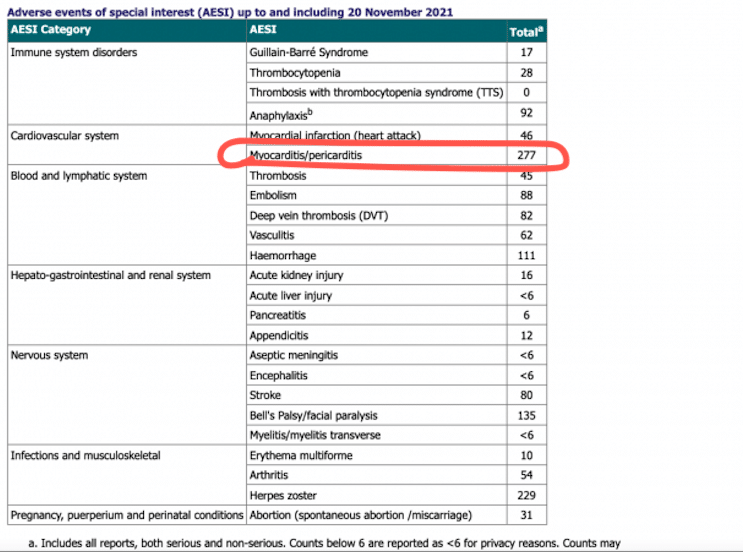
This monitoring strategy is continuous, proactive and based on a wide range of information sources, with a dedicated team of scientists reviewing information to look for safety issues or unexpected rare events. This is a requirement for all authorised medicines and vaccines in the UK. The clinical trials of COVID-19 vaccines have shown them to be effective and acceptably safe however, as part of its statutory functions, the MHRA is responsible for monitoring these vaccines on an ongoing basis to ensure their benefits continue to outweigh any risks. In relation to COVID-19 vaccines, the MHRA has authorised their supply following a rigorous review of their safety, quality and efficacy. The MHRA has played an active role in responding to the coronavirus pandemic. We would ask anyone who suspects they have experienced a side effect linked with their COVID-19 vaccine to report via the Yellow Card website. Additionally, monthly updates of Adverse Drug Reaction (ADR) data will continue with the new interactive COVID-19 vaccine reports. Robust safety monitoring and surveillance of any COVID-19 vaccines used in the UK will continue along with timely communication on any updated safety advice when needed. The report published 8 March 2023 is therefore the last regular publication of the Summary of Yellow Card reporting for COVID-19 vaccines. The Commission on Human Medicine (CHM) has advised that given the end of the Autumn 2022 booster campaign and the stable safety profile of the COVID-19 vaccines, the MHRA should transition to routine data publication and communication of safety concerns for COVID-19 vaccines. 
This report covers the period up to and including 22 February for COVID-19 vaccines used from the beginning of Autumn 2022.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
